Market Overview
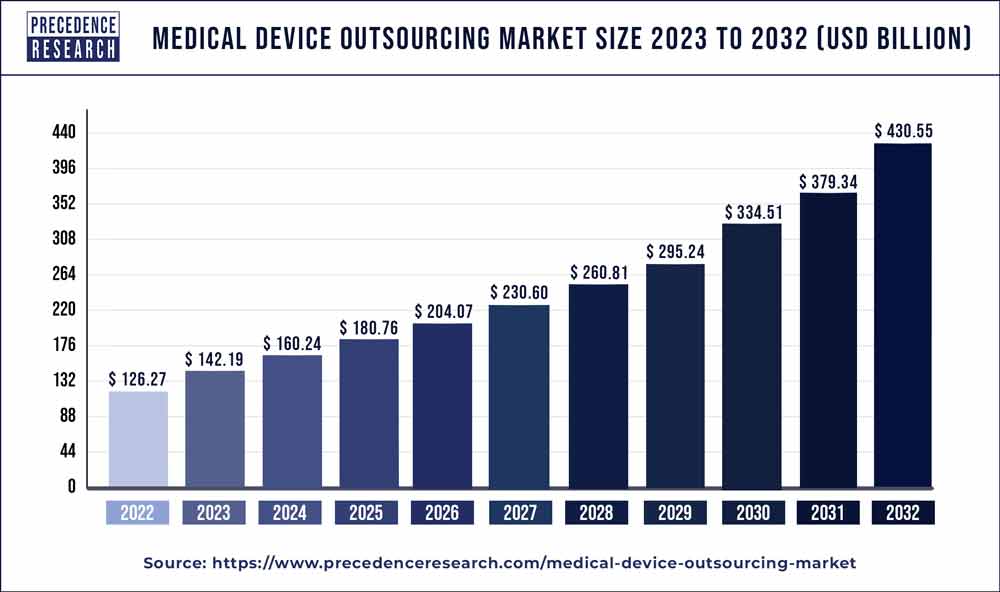
The global medical device outsourcing market has been witnessing significant growth over the past few years, driven by the increasing need for cost efficiency and swift production timelines. In 2023, the market was valued at USD 142.19 billion and is projected to reach USD 430.55 billion by 2032, expanding at a compound annual growth rate (CAGR) of 13.10%.
Key Highlights
- The Asia Pacific region held the biggest chunk of the market in 2023, at around 41.68%.
- North America is expected to see the fastest growth rate (CAGR of 12.56%) between 2023 and 2032.
- In terms of services, quality assurance was a major player in 2023, accounting for roughly 9.42% of the market.
- Regulatory affairs services are forecast to experience the most significant growth, with a CAGR of 13.54% projected from 2023 to 2032.
- As for applications, the drug delivery segment was a key contributor in 2023, holding around 20.83% of the market share.
- The ophthalmic segment is anticipated to have the most remarkable growth, with a CAGR of 13.39% estimated between 2023 and 2032.
Get a Sample: https://www.precedenceresearch.com/sample/1057
Key Market Dynamics
Drivers:
One of the primary drivers of the medical device outsourcing market is the increasing complexity of medical devices and technologies. As devices become more advanced, manufacturers often seek specialized expertise and capabilities that outsourcing partners can provide. This trend is further bolstered by the need for faster time-to-market and cost-efficiency in product development. Moreover, regulatory pressures and compliance requirements also drive companies to outsource to ensure adherence to global standards and regulations.
Restraints:
Despite its growth, the medical device outsourcing market faces certain restraints. One significant challenge is the risk associated with intellectual property (IP) protection and data security. Manufacturers must carefully navigate these concerns when outsourcing critical aspects of their product development and manufacturing processes. Additionally, quality control and supply chain risks can arise when relying on external partners, potentially impacting product reliability and market acceptance.
Opportunities:
Opportunities in the medical device outsourcing market are abundant, particularly in emerging markets and for niche technologies. Outsourcing allows companies to leverage geographical advantages, such as lower labor costs and proximity to target markets, thereby enhancing competitiveness. Furthermore, partnerships with outsourcing firms can facilitate access to advanced technologies and specialized skills, fostering innovation and differentiation in product offerings. Strategic collaborations also enable manufacturers to focus on core competencies while mitigating operational risks and capital expenditures.
Read Report: Analgesics Market Size to Reach USD 132.51 Bn By 2032
Regional Insights
Asia Pacific
In 2023, the Asia Pacific region asserted its dominance in the global medical device market, capturing a commanding 41.68% share. This leadership position is poised to persist, underpinned by robust growth projections. The market in this region was valued at USD 52.5 billion in 2023 and is forecasted to soar to USD 183.65 billion by 2032, marking a substantial compound annual growth rate (CAGR) of 13.39%. Several factors drive this exceptional growth trajectory.
Firstly, Asia Pacific benefits from a large and expanding patient pool grappling with prevalent neurological and cardiac disorders. This demographic imperative not only propels demand for medical devices but also incentivizes investments in advanced healthcare technologies to meet diagnostic and therapeutic needs.
Secondly, the region enjoys significant cost advantages. Lower labor costs and operational overhead compared to Western counterparts bolster manufacturing efficiencies and competitiveness. This cost-effectiveness is particularly attractive to multinational corporations seeking to optimize production and enhance profit margins.
Lastly, Asia Pacific has witnessed remarkable advancements in technical expertise and manufacturing capabilities. Investments in research and development, coupled with improvements in regulatory frameworks, have fostered an environment conducive to innovation. This technical prowess not only supports local market expansion but also positions the region as a pivotal player in the global medical device landscape.
North America
North America, particularly the United States, is set to experience robust growth in the medical device market, projected to expand at a CAGR of 12.56% from 2023 to 2032. This accelerated growth trajectory is underpinned by several key drivers.
Foremost among these drivers is the high demand for advanced medical devices driven by a sophisticated healthcare system and a culture of innovation. The United States, home to a thriving medical device industry, continually pushes the boundaries of technology and therapeutic interventions. This demand is fueled by a healthcare ecosystem that prioritizes cutting-edge solutions to address complex medical challenges.
Additionally, North America benefits from a favorable regulatory environment that supports market expansion. Stringent yet predictable regulatory frameworks provide clarity and stability for manufacturers and investors alike. This regulatory support, combined with a well-established healthcare infrastructure, facilitates rapid adoption of new medical technologies and treatments.
Service Insights
The market is segmented by various services, including:
- Quality Assurance: Contributing 9.42% to the market in 2023, this segment is crucial for ensuring compliance with regulatory standards.
- Regulatory Affairs Services: Expected to grow at a CAGR of 13.54%, driven by the increasing complexity of global regulatory requirements.
- Contract Manufacturing: This segment held the largest revenue share in 2022 and is anticipated to maintain its dominance, driven by the need to reduce production costs.
Application Insights
The market is also segmented by application, with notable segments including:
- Drug Delivery: Contributed 20.83% in 2023, reflecting the high demand for efficient drug delivery systems.
- Ophthalmic: Expected to grow at a CAGR of 13.39%, driven by advancements in eye care technologies.
- Cardiology: Estimated to record the fastest growth due to the increasing prevalence of cardiovascular diseases.
Competitive Landscape
Key players in the market include:
- Intertek Group PLC
- TüvSüd AG
- Wuxi Apptec
- SGS SA
- Toxikon, Inc.
- Eurofins Scientific
Segments Covered in the Report
By Service
- Product Upgrade Services
- Regulatory Affairs Services
- Legal representation
- Clinical trials applications
- Regulatory writing and publishing
- Quality Assurance
- Product Maintenance Services
- Product Testing & Sterilization Services
- Product Design and Development Services
- Molding
- Designing & engineering
- Machining
- Packaging
- Product Implementation Services
- Contract Manufacturing
- Accessories manufacturing
- Component manufacturing
- Device manufacturing
- Assembly manufacturing
By Application
- Drug delivery
- Dental
- Diabetes care
- Cardiology
- Endoscopy
- IVD
- Ophthalmic
- Diagnostic imaging
- Orthopedic
- General and plastic surgery
- Others
By Regional
- North America
- U.S.
- Canada
- Europe
- U.K.
- Germany
- France
- Asia Pacific
- China
- India
- Japan
- South Korea
- Middle East & Africa
- Latin America
Immediate Delivery Available | Buy This Premium Research Report@ https://www.precedenceresearch.com/checkout/1057
